 
In covalent bonds, two elements will share valence electrons until they have a complete shell. One of the primary characteristics of nonmetals is that they form chemical compounds by making covalent and ionic bonds. The noble gases are:ĥ Properties Of Nonmetals 1. As such, noble gases typically do not react with other elements to form compounds. Noble gases have a full valence shell of 8 electrons so they exist in a very stable electron configuration. In contrast, the noble gases are characterized by their inert chemical behavior. All halogens have a valence shell of 7 electrons so they are extremely reactive and readily form negative ions. 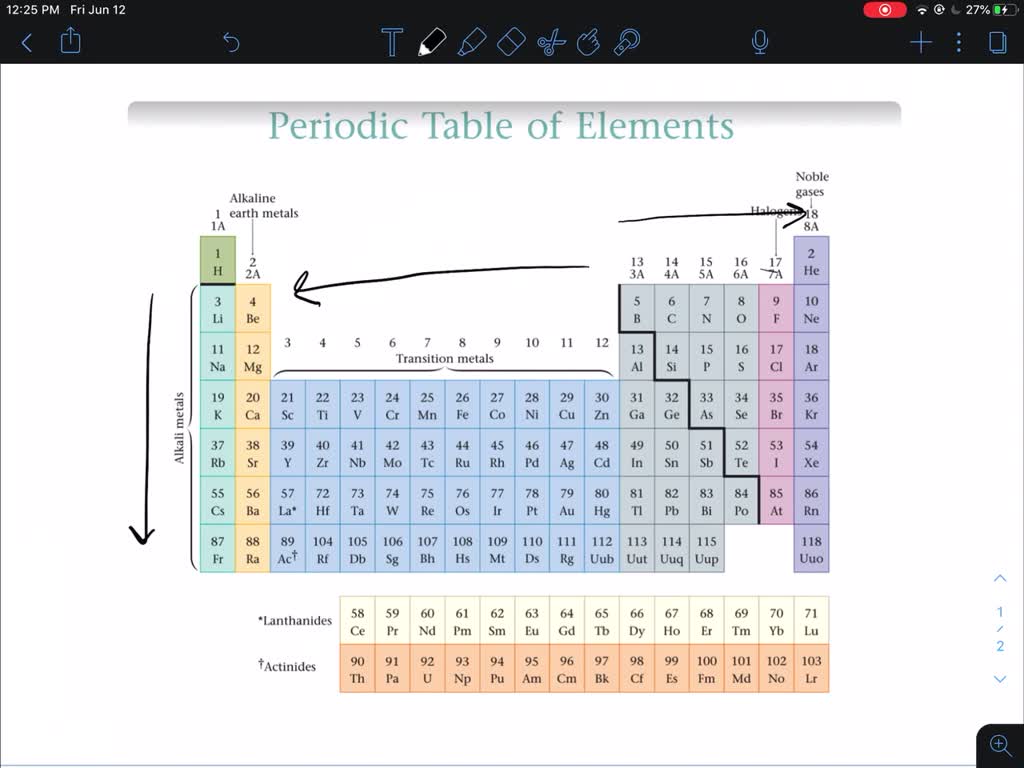
The reactive nonmetals also include the halogens-elements in group 17 of the periodic tables. Others, like oxygen, are a gas at room temperature and are very electronegative. Some, like carbon and sulfur, are solid at room temperature and are less electronegative. Reactive nonmetals tend to show more variation in their physical and chemical properties. Generally, nonmetals are split into two categories: reactive nonmetals and halogens. Polymers such as plastic are covalently bonded chains of molecules that are malleable and can be shaped. Graphite, for example, is a nonmetal compound that is a very good conductor of electricity and is used in electronics equipment. Some nonmetals have all of these properties, some have very few. This list of properties is neither essential nor exhaustive.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
